Uganda develops own COVID-19 vaccine, asks citizens to volunteer for trials
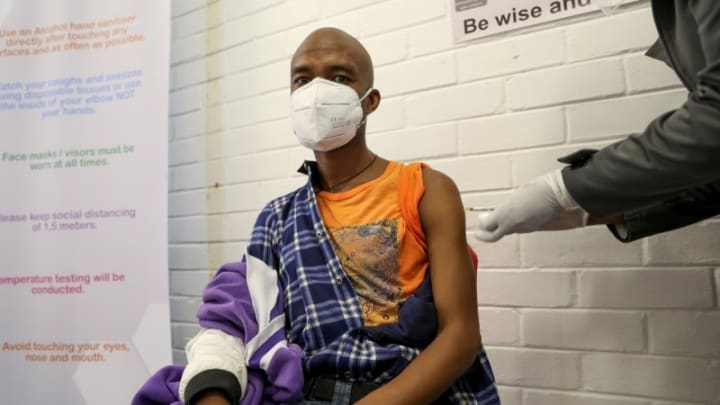
Uganda develops own COVID-19 vaccine, asks citizens to volunteer for trials
Uganda has said its scientists and researchers have developed a scientifically proven vaccine that will be used for coronavirus treatment after trials.
The Presidential Scientific Initiative on Epidermic (PRESIDE) in a statement on Wednesday, January 27, said a team a from Natural Research Centre began COVID-19 research in April 2020 following government approval.
As a result, the team that was led by Grace Nambatya Kyeyune developed a product code UBV-01N whose trial is expected to validate its content standardisation and replication.
“Today, we are witnessing the first clinical trial of a highly scientific process to analyse and validate a natural product anticipated to have anti-viral and immunological ability,” stated PRESIDE.
Phase one of the trial will involve 128 adult volunteers aged between 18-70 and must be positive for coronavirus by the time of trial. Members of the public have been urged to willingly take part in the exercise as the East African country race against time to beat the viral infection from claiming more lives. Gov’t says plan to buy hospital beds from local artisan flopped because supplier was crime suspect
“We appeal to Ugandans to welcome the clinical trial and willingly participate in when to volunteer. The product has been proven safe and the trial is basically to prove its efficacy against COVID-19,” PRESIDE added.
Since March 2020, Uganda has recorded 39, 261 positive cases out of which 318 patients were confirmed dead from the infection as on Wednesday, January 27. In terms of recovery, 14,052 patients have so far been discharged from various health facilities from the total 820, 154 cumulative cases. Elsewhere in Kenya, Health Cabinet Secretary Mutahi Kagwe said the country could get its dose of COVID-19 vaccines it had ordered by February 2021 or “even earlier”. Kagwe, who addressed a pressed conference at Kenyatta Univesity (KU) on Wednesday, January 13, said plans to avail the vaccines in the country were at an advanced stage. Kagwe, who did not give more details concerning the jab that will be deployed in the country, disclosed that Kenya will be using more than one vaccine to combat the disease. “We have engaged various institutions, notably GAVI, the organisation leading in the acquisition of vaccines expected in Kenya in February. However, it is even possible to get the vaccines in Kenya earlier than that. The arrangements being made by both public and private sector are likely to see the vaccine here before the February date given by GAVI,” said Kagwe





